
Two Lines of Research
The Fitch lab has two main lines of research that it conducts. Both focus on developmental disorders and disruptions that occur in humans, and modeling those in rodents. Read about each below.
Hypoxic-Ischemic Brain Injury and Behavioral Outcome
Infants born prior to 38 weeks gestation or less than 3 pounds (very low birth weight (VLBW)) are at an increased risk for intraventricular and periventricular hemorrhage (IVH-PVH; these early injuries are typically diagnosed by bed-side ultrasound in the NICU) and hypoxic-ischemic (HI) damage. Children born prematurely or VLBW also have an elevated risk for later cognitive and language disabilities. Similarly, infants who experience trauma in the birth process (prolonged labor, cord prolapse, placenta previa) may experience HI episodes leading to brain injury. Evidence suggests that the hypoxic-ischemic tissue loss resulting from such injuries may lead directly to subsequent behavioral deficits. By inducing comparable hypoxic-ischemic injuries in perinatal rats, we can study issues including long-term behavioral outcome as a function of parameters such as timing and severity of injury, as well as the role of gender, and the efficacy of various neuroprotectants for improving long-term cognitive outcome.
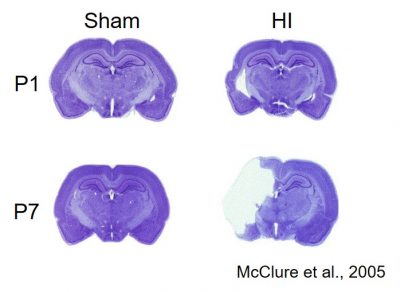
Cross-section of rat brains showing tissue loss following HI Injury at 1 day old (P1) and 7 days old (P7).
Genetic Models of Language Related Developmental Disorders
The role of genetics in language has gained particular attention, given accumulating knowledge about the genes, proteins, and cellular machinery involved in both brain development and cognitive outcomes. Disruptions to various neurodevelopmental mechanisms (e.g. neuronal migration and synaptic plasticity) have been related to impairments in language as seen in neurodevelopmental disorders, including dyslexia, specific language impairment (SLI), and autism spectrum disorders (ASDs). Current research in the lab targets specific genes implicated in various language-related disorders, and assesses the behavioral and neuroanatomical consequences of altering the protein products associated with these genes, via the use of transgenic mouse models. We can explore the gene-brain-behavior relationship to further examine how each targeted gene mediates core behavioral endophenotypes associated with language and reading-relation function. Our novel behavioral paradigms can tap into these “intermediate” language-related behavioral phenotypes in mice, which include: rapid auditory processing, visual motion perception, sensorimotor functions, social/communicative interactions, and working memory. Additionally, we can quantify gross neuroanatomy to assess whether neural anomalies correlate with any atypical behavior. Future applications of data could allow for more precise diagnosis, and advanced early screening tools as well as more targeted interventions using these genetic and anatomical markers.
Ongoing Research:
- USH2A and Usher Syndrome Type II:
- Studying the role of USH2A and Developmental Language Delay
- UBE3A and Angelman Syndrome:
- A behavioral and neuroanatomical characterization of Ube3a KO mice
- DCDC2 and KIAA0319:
- Studying the role of dyslexia-risk genes and visual motion perception
Behavioral Testing
Rapid Auditory Processing
We use a modified non-invasive “startle reduction” behavioral paradigm to assess how well subjects rapidly discriminate complex sounds (similar to components of human speech). Animals must be able to detect an auditory cue that indicates a startle eliciting stimulus (SES) is about to occur. If the animal is able to detect and process the cue, they will have a lowered startle response than if no cue was given.
Learning and memory
We can also assess subjects on a variety of learning and memory tasks, such as spatial maze navigation, and operant conditioning.

A rat swims in a Morris water maze to find a hidden platform (Jean-Etienne Poirrier, 2007)
Social Behavior
An important factor in many developmental disruptions and disorders, we can evaluate an animal’s social performance in several different situations. Interest in social opportunities can be tested using a 3-chamber social choice task. Social aggression and dominance is evaluated on a tube task. Ability to communicate is measured by recording vocalizations during rodent interactions.

A mouse navigates a 3-chamber social choice task